
KINGSTON, R.I. — April 23, 2026 — In 2024, Assistant Professor Yang Lin was awarded the Defense Advanced Research Projects Agency Young Faculty Award, a highly competitive program supporting early career researchers pursuing high-risk, high-impact research. The award provided two-year funding, mentorship, and access to Department of Defense and industry networks to support his project developing a next generation microphysiological system called the Precision Lattice Acoustofluidic Tissue Ecosystem. That project was recently extended for a third year of funding under the agency’s prestigious Director’s Fellowship Award, granted to roughly 10 top-performing projects annually from the DARPA Young Faculty Award pool.
Lin is the first University of Rhode Island faculty member to receive the DARPA Young Faculty Award and the first to be selected for the highly competitive Director’s Fellowship, marking a significant milestone in his career. The additional third year will provide $300,000 in funding, bringing the total funding for the project to approximately $800,000.
“I am deeply honored to receive the DARPA director’s fellowship,” said Lin. “What makes this recognition especially meaningful is that it reflects not just the promise of an idea, but the progress we have already demonstrated and the direction this work is now taking. It provides an opportunity to move beyond initial demonstrations and push toward more integrated and transformative systems.”
The success of the initial research yielded findings that demonstrate a new approach to addressing one of the fundamental challenges in engineered tissue systems: how fluids, nutrients, and biochemical signals are transported within three-dimensional structures. In most current organoid and tissue-chip platforms, transport is governed by passive diffusion, which becomes increasingly limiting as systems grow in both size and complexity. As a result, system behavior is often shaped by transport constraints rather than intrinsic biological processes, limiting both scalability and interpretability.
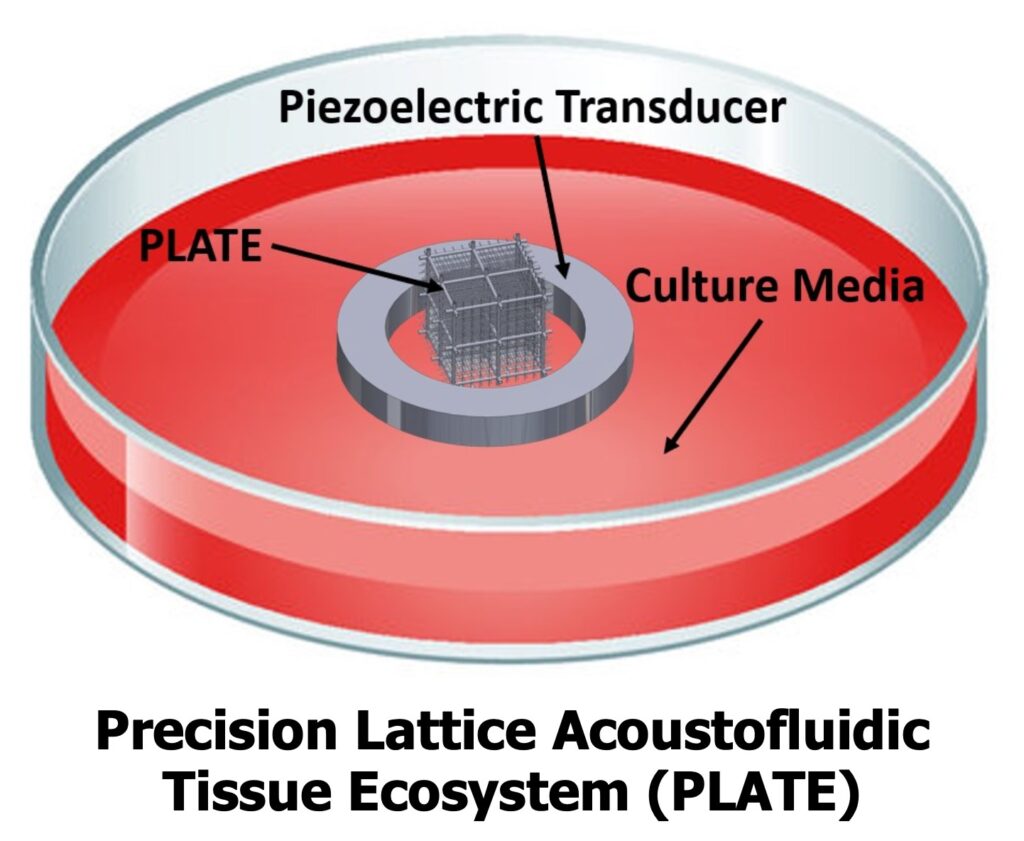
Lin’s research introduces an alternative approach through the PLATE platform, which integrates acoustofluidic elements directly into three-dimensional tissue architectures. By using acoustically-driven flow, the system enables programmable transport within the internal volume of engineered tissues, rather than relying on passive processes at the boundaries. This enables more uniform nutrient delivery, improved removal of metabolic waste, and precise control over spatial gradients, all of which are critical for maintaining viable and functional tissue systems.
“I believe this work has the potential to change how the field approaches engineered tissues at a fundamental level,” said Lin. “Today, even the most advanced organoid and tissue-chip systems remain constrained by transport limitations.”

More broadly, this approach allows transport to be treated as a controllable design variable, rather than a limiting constraint. This shift has the potential to significantly improve the reliability and scalability of organoid and tissue-chip models, and to enable new experimental capabilities in studying complex biological systems. In practical terms, that could make these systems more useful across a range of biomedical applications. For example, they could help researchers build more realistic laboratory models for studying disease and evaluating potential therapies in systems that behave more like real human biology. They could also support the development of next-generation engineered tissues by providing more precise control over how nutrients, oxygen, and biochemical signals move through three-dimensional structures. Over time, advances like this could generate more reliable results earlier in the development process and reduce reliance on less predictive testing approaches.
The director’s fellowship supports the next stage of this work, moving from initial feasibility demonstrations toward fully integrated microphysiological systems in which fluid flow, nutrient delivery, and spatial gradients can be engineered with precision and coordinated with biological function. In this next phase, the work will focus in part on applications in complex 3D tissue systems, including brain organoids, where controlled transport is critical for maintaining viability and function, and will be further enabled through collaborations in organoid and tissue engineering. The broader goal is to establish a new framework in which transport and biology are co-designed, enabling systems that are both more physiologically relevant and more experimentally controllable.
Lin joined URI’s College of Engineering Mechanical, and Industrial Systems department in August 2020 after completing his Ph.D. in mechanical engineering at the University of Illinois Chicago in December 2019. His research focuses on microfluidics and acoustofluidics, microphysiological systems, organoid engineering, acoustic metamaterials, environmental sensing of microplastics and PFAS, and advanced microfabrication and 3D printing.
He is also the recipient of a 2026 National Science Foundation CAREER award and a 2024 NSF Engineering Research Initiation award.
This story was written by Krysta Murray, writer, College of Engineering.
